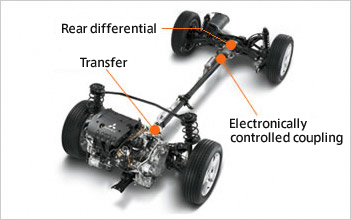
[image_frame style=”framed_shadow” align=”right” alt=”e-fuel injection in glass engine” title=”Precision injection – just a few milliliters of fuel at a time are injected into the pressure chamber.” height=”200″ width=”150″]https://www.car-engineer.com/wp-content/uploads/2014/02/e-fuel-injection-in-glass-engine.jpg[/image_frame]The development of Audi e-fuels has reached a new milestone. The synthetic fuels have now completed a test cycle in Ingolstadt in the pressure chamber and the glass engine – with the following results: Audi e-fuels often perform better than their conventional counterparts.
“Our test shows that as well as electric driving on renewable electricity, there are other concepts that permit long-distance, low-emission driving,” commented Reiner Mangold, Head of Sustainable Product Development at Audi.
The engineers were first and foremost interested in mixture preparation and the synthetic fuels’ combustion characteristics. To investigate these, they simulated the conditions inside an engine in a pressure chamber at up to 15 bar and temperatures of 350 degrees Celsius. A special camera recorded how the fuel behaves during the injection process, because only a clean mixture preparation process will assure optimal combustion.[image_frame style=”framed_shadow” align=”center” alt=”Fuel sprayed directly into the cylinder” title=”Left to right: When the intake valves are open, the fuel is sprayed directly into the cylinder, where it distributes evenly. Combustion is triggered by the ignition sparks, from where the flame front propagates. The increasing pressure generated by combustion pushes the piston downward.” height=”198″ width=”600″]https://www.car-engineer.com/wp-content/uploads/2014/02/Fuel-sprayed-directly-into-the-cylinder.jpg[/image_frame]
The glass engine was another test station. This test setup reveals the processes that are otherwise hidden by the metal walls of the cylinders. A small window made of quartz glass enables the experts to observe the fuel’s behavior in the cylinder and how it interacts with the airflow in the combustion chamber. During each of up to 3,000 revolutions per minute in the research engine, a minute amount of fuel shoots into the glass cylinder, is compressed and ignited, and the exhaust gas then expelled.
Using a high-speed camera, the combustion process is captured with time-lapse photography. “We examine where and how the fuel ignites in the cylinder,” says Peter Senft, thermodynamics expert. “The blue flame is an indicator that the fuel has been cleanly and fully combusted.” But it doesn’t end there. In contrast to fossil fuels, which have varying compositions depending on their geographical source, Audi e-fuels are absolutely pure. Peter Senft explains, “Thanks to their chemical characteristics, they generate fewer pollutants during combustion. They contain no olefins and no aromatic hydrocarbons.” To sum up – better mixture formation, cleaner combustion and fewer emissions.[image_frame style=”framed_shadow” align=”center” alt=”Behavior of the fuel as it is injected into the pressure chamber” title=”Left: The experts from Technical Development use light scattering to analyze the behavior of the fuel as it is injected into the pressure chamber. Right: They then send a laser beam through the fuel spray to investigate its inner workings.” height=”300″ width=”600″]https://www.car-engineer.com/wp-content/uploads/2014/02/Behavior-of-the-fuel-as-it-is-injected-into-the-pressure-chamber.jpg[/image_frame]
The next task already awaits Audi’s engineers: They now aim to further optimize the production process for e-ethanol and e-diesel. These new fuels will then be ready for the market. Audi operates a research facility in Hobbs (New Mexico, United States) for the production of e-ethanol and e-diesel in partnership with Joule. At this facility, microorganisms use water (brackish, salt or wastewater), sunlight and carbon dioxide to produce high-purity fuels. The e-gas project in Werlte is another component of Audi’s extensive e-fuels strategy.
Source: Audi
[titled_box title=”Romain’s opinion:”]
Synthetic fuels are often very convenient from a combustion point of view as they don’t contain sulfur, and other aromatics. However, the carbon balance is not that good when we take into account the difficulty to produce it. It is indeed very demanding in terms of energy to create synthetic fuels like these ones. And depending on the energy mix of the country where it is produced, it can end up to an even worse carbon balance than with conventional fuels. Do you think that these synthetic fuels will anyway arrive in the market? What about the storage properties of these fuels?[/titled_box]